Appoint Your EU Authorised Representative in Just 24 Hours
We deliver professional UK and EU Authorised Representative services for various product sectors, including consumer goods, electronic devices, toys, cosmetics, medical devices, and in vitro diagnostic products (IVDs). Our system also supports the creation of Declarations of Conformity and helps maintain full regulatory compliance within European markets.

Trusted by Leading Brands

All-in-One Product Compliance Support
Rapid EU Authorised Representative Appointment
If your business is not legally based in the EU, numerous regulations — including the Toy Safety Directive, MDR, IVDR, EMC, LVD, RED, PPE Regulation, and Cosmetics Regulation — require you to designate an EU Authorised Representative. We supply a confirmed EU address within 24 hours, allowing you to quickly update product labelling, packaging details, technical documentation, and online marketplace information to meet legal requirements.


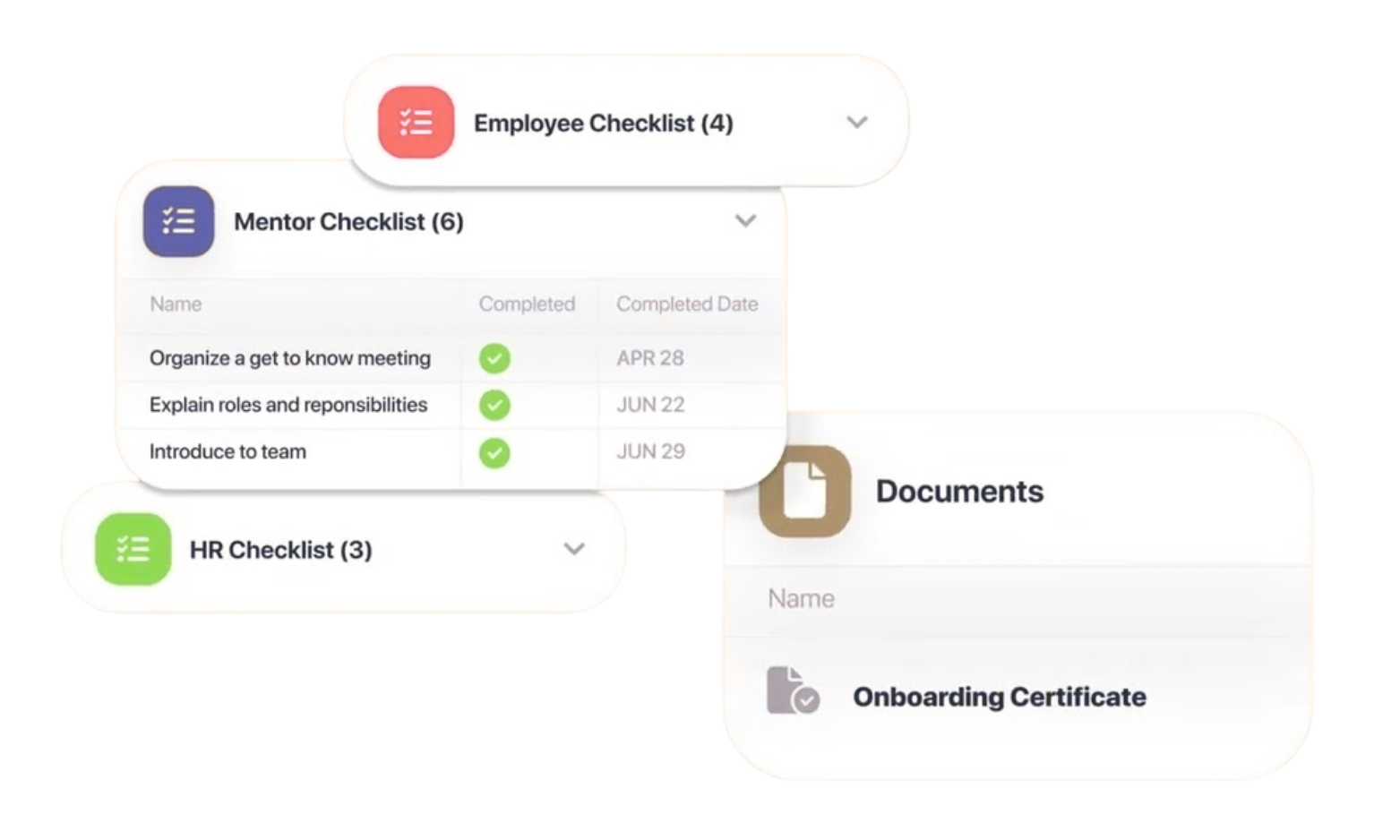
Smart Technical File Management
Register your products through a straightforward guided process. Our platform determines the relevant EU directives and regulations applicable to your product and assists you in compiling, organising, and securely storing your complete technical file in one secure location.
Step-by-Step Risk Assessment Framework
Most EU and UK product laws mandate a documented risk assessment. Our structured template helps you identify potential hazards, evaluate risk levels, and establish appropriate control measures to satisfy compliance obligations.


E-Commerce Compliance Assistance (Amazon, Etsy, eBay, Shopify)
Ensure your product listings comply with safety and regulatory standards. Our platform enables you to create GPSR-compliant documentation — including conformity declarations, certificates, and laboratory reports — helping your listings remain approved and active across major online marketplaces.
10-Year Record Retention & Regulatory Communication
Under GPSR rules, product safety records must be maintained for up to 10 years. We securely archive your technical documentation for the required timeframe and provide assistance in responding to EU Market Surveillance Authority enquiries, including support in relevant local languages when necessary.


Recall Guidance & EU Safety Business Gateway Filing
In the case of a product safety concern, EU legislation requires formal recall and reporting procedures. We support you throughout the recall process and assist with submitting the necessary notifications through the EU Safety Business Gateway, ensuring compliance with legal obligations.
Pay by Product Type — Not Individual Variants
For most products, compliance under GPSR is based on the product type rather than each individual variation such as size, colour, or model. This means you only need to register the product once, reducing unnecessary SKU-level submissions and improving efficiency. (Some regulated product categories, such as toys, may still require documentation for each individual SKU.)

Key Compliance Steps
Start your free trial and complete your manufacturer profile.
Provide your company and manufacturing details to get started.
Evaluate your product, document compliance, assess risks, and meet GPSR and other regulatory requirements.
Add mandatory markings and warnings such as CE, UKCA, and EU REP to your product and packaging.
Add your products, identify applicable regulations, and ensure ongoing compliance.
Upload technical files, generate Declarations of Conformity, and demonstrate regulatory compliance.
Who Needs EU
Authorised Representatives?
- Manufacturers
- Importers
- Amazon sellers
- Etsy sellers
- Shopify sellers
- Private label brands
- Artists, crafters, book creators
- Home & lifestyle brands
- Electronics & gadget brands
Why Choose Complico?
EU Authorised Representative services activated within 24 hours
An all-in-one system for technical documentation, risk evaluation, and product traceability
Smart, AI-powered assistance for faster and more accurate document creation
EU Authorised Representative services activated within 24 hours

Schedule a Discovery Call
Not sure how to begin with GPSR compliance?
Chat with our specialists to learn what’s needed for EU & UK markets and how Complico Consulting makes compliance straightforward.
Frequently asked questions
An EU Authorised Representative (EU AR) is a company officially designated within the European Union to act on behalf of a manufacturer located outside the EU. Their role is to represent the manufacturer in relation to regulatory responsibilities for products placed on the EU market.
You are required to appoint an EU Authorised Representative if your company is established outside the European Union and you intend to sell products in the EU that are subject to specific European directives or regulations. These may include frameworks such as the General Product Safety Regulation (GPSR), Medical Devices Regulation (MDR), In Vitro Diagnostic Regulation (IVDR), Toy Safety Directive, EMC Directive, Radio Equipment Directive (RED), Low Voltage Directive (LVD), PPE Regulation, Machinery Regulation, Cosmetics Regulation, RoHS Directive, and other applicable EU legislation.
An EU Authorised Representative is generally required for products that fall under EU regulatory controls. Common product groups include:
Electrical and electronic devices
Toys and products intended for children
Cosmetic and beauty items
Medical devices and in vitro diagnostic products (IVDs)
Industrial equipment and machinery
Personal protective equipment (PPE) and safety gear
Radio equipment, IoT devices, and wireless technologies
Materials intended for food contact
General household and consumer products
Chemical substances and aerosol products
If the applicable EU legislation requires a representative established within the European Union, one must be formally appointed before the product can be legally placed on the EU market.
An EU Authorised Representative (AR) or Responsible Person (RP) is responsible for carrying out key regulatory functions on behalf of a non-EU manufacturer. Their obligations typically include:
Retaining and managing your technical documentation
Confirming that the product complies with all relevant regulations before it is placed on the EU market
Serving as the official point of contact for regulatory authorities
Supporting and coordinating inspections, compliance checks, and testing requests
Managing product safety issues, including recalls and required notifications through systems such as SCMS or the Safety Business Gateway (SBG)
Verifying that product labelling, CE or UKCA markings, and traceability details are accurate and compliant
Keeping regulatory documentation on file for the legally required retention period, typically between 10 and 15 years depending on the applicable legislation
The requirement to designate an EU Authorised Representative depends on the type of product being placed on the market. Various EU laws and directives may mandate the appointment of a representative established within the European Union, including:
General Product Safety Regulation (EU 2023/988)
Toy Safety Directive (2009/48/EC)
Medical Devices Regulation (EU 2017/745)
In Vitro Diagnostic Regulation (EU 2017/746)
Electromagnetic Compatibility (EMC) Directive (2014/30/EU)
Low Voltage Directive (2014/35/EU)
Radio Equipment Directive (2014/53/EU)
Personal Protective Equipment Regulation (EU 2016/425)
Machinery Regulation (EU 2023/1230)
Cosmetics Regulation (EC 1223/2009)
Restriction of Hazardous Substances (RoHS) Directive (2011/65/EU)
REACH Regulation (EC 1907/2006)
Food Contact Materials legislation
The specific requirement will depend on your product’s classification and the applicable legal framework.
Euverify provides support across the principal EU and UK compliance regimes to help ensure your products meet all relevant regulatory obligations.
Yes, it is still required.
Selling products online is legally treated in the same way as selling through physical retail channels. E-commerce platforms and online marketplaces typically require a valid EU-based contact address and may request details of your Authorised Representative or Responsible Person, along with relevant certifications, declarations, and product safety documentation before allowing products to remain listed.
The documents needed will vary depending on your product category, but may include:
Complete technical documentation file
EU Declaration of Conformity
Relevant test reports in line with applicable EN or ISO standards
A documented risk assessment
Details of your manufacturing process and facility
Final product label artwork showing the AR/RP address
Safety Data Sheets (for chemicals or aerosol products)
Product Information File (PIF) and Cosmetic Product Safety Report (CPSR) for cosmetics
Regulatory and technical documentation for medical devices
Complico Consulting provides structured, step-by-step guidance to help you compile and submit the correct documentation for your specific product type.
In most cases, your EU Authorised Representative service is activated within 24 hours once the following steps have been completed:
Account registration is finalised
Your company information has been provided
The digital Authorised Representative agreement has been signed
All required documentation has been submitted
Once these requirements are met, activation is typically processed within one working day.
Your product labelling will include the following details:
The EU Authorised Representative address located in Ireland
The UK Responsible Person address based in England
These addresses are provided exclusively to registered Euverify clients and may be used on your packaging, labelling, and related compliance documentation.
Yes.
All submitted documentation is securely archived for the full legally required retention period, typically between 10 and 15 years depending on the applicable regulation.
Regulatory authorities may request access to these records at any time, and Euverify handles all official correspondence and communication with the relevant authorities on your behalf.
Yes.
Your technical file is carefully evaluated to ensure it is complete, properly structured, and aligned with applicable regulatory requirements. If any information or documentation is missing, this will be highlighted, and clear guidance will be provided on what needs to be corrected or added.
If a regulatory authority makes contact, our compliance specialists will manage the matter on your behalf. This includes:
Communicating directly with the relevant authority
Supplying requested technical documentation
Coordinating and managing compliance or safety verifications
Advising you on any required corrective measures
Assisting with product recalls or mandatory incident reporting, where applicable
Throughout the process, you will be kept fully updated and informed of any developments.
Yes.
Euverify can act as your EU Authorised Representative for Class I medical devices and provides support with key regulatory obligations, including:
EUDAMED registration procedures
Manufacturer registration requirements
UDI implementation and product traceability
Review and verification of technical documentation
Management of safety-related communications
Dedicated Authorised Representative services for medical and IVD products are available to ensure compliance with the relevant EU regulations.
Yes.
Euverify can act as your Responsible Person for cosmetic products placed on the EU market. Our services include:
Preparation and review of the Cosmetic Product Safety Report (CPSR)
Compilation and maintenance of the Product Information File (PIF)
Submission of product details through the Cosmetic Products Notification Portal (CPNP)
Compliance review of product labelling
Management of required safety documentation
Ongoing post-market surveillance support
We help ensure your cosmetic products meet all regulatory obligations before and after they are made available within the European Union.
CE marking is required for products placed on the European Union market. It indicates that the product complies with applicable EU legislation and regulatory requirements.
UKCA marking (UK Conformity Assessed) is required for products sold in Great Britain (England, Scotland, and Wales). It confirms compliance with relevant UK regulations following Brexit.
Depending on where your products are distributed, you may need one marking or both to legally place your goods on the respective markets.
Euverify provides guidance and support for both EU and UK compliance pathways to help ensure your products meet all applicable requirements.
If you’d like, I can also provide a more technical regulatory explanation or a simple comparison table format for your website.
Can I appoint Euverify solely for the Authorised Representative address without using the platform?
Can Complico Consulting serve as Authorised Representative for more than one brand or product range?
Yes.
Our service is designed to accommodate multiple brands, product categories, and product lines under a single account. This allows you to manage compliance for different product families efficiently through one central arrangement.
You are required to inform us of any changes.
Depending on the nature of the update, it may be necessary to submit revised technical documentation or provide an updated technical file to ensure continued regulatory compliance.
Yes — in fact, this is precisely the purpose of Authorised Representative (AR) and Responsible Person (RP) services.
We work with manufacturers and businesses located outside the European Union, including companies based in the United States, China, Switzerland, the UAE, Australia, India, and many other countries worldwide.
No.
There is no long-term commitment required. You may cancel your subscription at any time, and our services do not include mandatory minimum-term agreements.
Yes.
Euverify provides support with product recall procedures and notifications through the Safety Gate (formerly known as RAPEX), helping ensure regulatory obligations are properly met.
You can choose the level of service that suits your business needs:
EU Authorised Representative service only
Combined EU and UK compliance support (recommended for brands operating internationally)
We help guide you through the required steps and manage communications where necessary to maintain compliance.
Euverify provides AR support across a broad range of regulated industries, including:
Consumer goods
Electrical and electronic products
Toys and children’s items
Cosmetic and beauty products
Medical devices
Personal protective equipment (PPE)
Food-contact materials
Machinery and mechanical equipment
DIY products and tools
Sports and outdoor equipment
Household goods
Industrial machinery and equipment
Our services are designed to accommodate diverse product categories while ensuring compliance with applicable EU and UK regulations.
Yes.
As part of our service, you are permitted to display our EU or UK address on your product packaging and labelling, where required by applicable regulations. This ensures your products include the legally mandated local contact details when placed on the relevant market.
All documentation is protected through encryption and stored within secure systems located in the European Union.
Access to files is strictly limited to authorised personnel only, ensuring controlled and secure handling of sensitive information.
Complico Consulting operates in full compliance with the requirements of the General Data Protection Regulation (GDPR), maintaining high standards of data privacy and security at all times.
